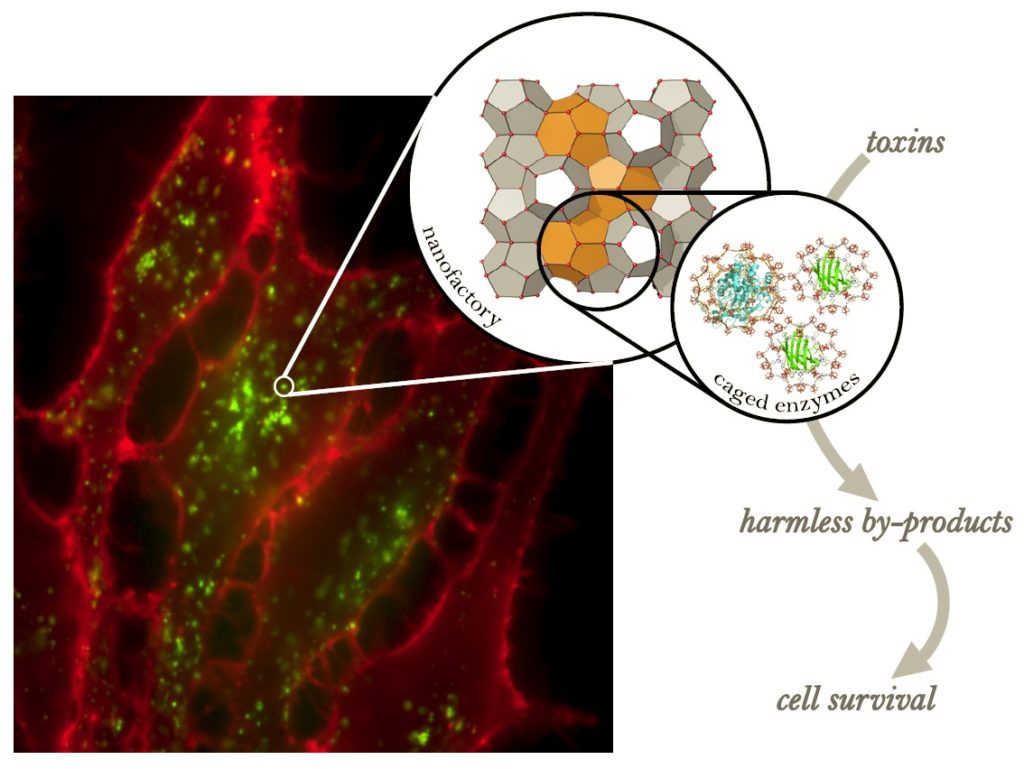
Two Texas A&M University laboratories have teamed up to create one small first with big potential beyond biochemistry circles — an enzymatic nanoreactor with the power to protect living cells from damage for more than a week.
The research, published earlier this month in the journal Nature Communications and funded in part by the Welch Foundation and the National Institutes of Health, is a collaborative effort between the labs of Texas A&M chemist and metal-organic framework (MOF) expert Hongcai Joe Zhou and Texas A&M AgriLife Research biochemist Jean-Philippe Pellois, who also holds a joint appointment in the Texas A&M Department of Chemistry.
By pairing Zhou’s talent in turning MOFs into molecular sieves capable of precisely separating targeted compounds with Pellois’ prowess in chemically manipulating protein structures inside living cells, the two labs successfully created customizable nanofactories — inorganic material-based nanocages that can encapsulate enzymes and offer unprecedented potential across a host of industries and disciplines.
“This research is at the interface of materials chemistry, cell biology and biomedical engineering,” said Texas A&M chemistry graduate student Xizhen Lian, a Zhou Laboratory member since 2013 and first author on the team’s paper. “The nanoreactor may be utilized in a number of biomedical applications, such as therapy for various diseases, from lysosomal storage disorders to cancer.”
Pellois notes that, when administered externally, enzymes typically are rapidly degraded by cells. In contrast, he says the semi-synthetic enzyme nanofactories the Texas A&M team has developed are extremely stable once inside cells. In addition, they are just as efficient and functional as the enzymes themselves, meaning that the caging process does not lead to a loss of activity.
“In this proof-of-concept study, we have shown that the nanofactories could be used to protect cells from harm as a result of a toxic pesticide or from reactive oxygen species,” Pellois added. “The protective effect provided by the nanofactories lasts for more than a week, which is in sharp contrast to the short-term protection effect — essentially a couple of hours — that is achieved with non-caged, non-protected enzymes.”
Pellois cites lysosomal storage disorders as one example of a disease in which a person’s cells either contain defective enzymes or don’t have the right ones present. One possible way to treat such diseases is to replace the defective enzymes by injecting purified enzymes into the patient — a previously problematic approach due to enzymes’ short half-lives.
“This type of approach requires frequent injections, something that is both very costly and unpleasant for patients,” Pellois said. “Our hope is that the new material we have developed could pave the way for enzyme replacement therapies that require less frequent injections.”
Beyond diseases and therapeutics, Pellois says scientists can begin thinking about the possibility of repurposing organelles and introducing new functions inside cells — pioneering steps that may prove useful in protecting cells ex vivo and in making cells resistant to stresses that may occur during a variety of procedures, such as transplantation, cultures and expansion. He said the team also hopes to test new enzymes that provide new functions and also whether the nanofactories show promise in animal models of disease.
The team’s paper, “High efficiency and long-term intracellular activity of an enzymatic nanofactory based on metal-organic frameworks,” can be viewed online along with related figures and captions.
To learn more about the team’s work, visit both the Pellois and Zhou research group websites.
###
Media contact: Shana K. Hutchins, (979) 862-1237 or shutchins@science.tamu.edu; Dr. Jean-Philippe Pellois, (979) 845-0101 or pellois@tamu.edu; or Dr. Hongcai Joe Zhou, (979) 845-4034 or zhou@chem.tamu.edu.





